These Shapes are always Polar Learn with flashcards, games, and more — for free.. How to use molecular geometry and Lewis structures along with VSEPR rules to ... Note: molecules with two atoms are not shown in these examples; they are always ... Electron Regions, shape, & hybridization, Bonding Regions, Lone Pairs ...
- what two shapes always produce polar molecules
- what shapes always yield polar molecules why
- what shapes produce polar molecules
http://purplebonding.comA polar molecule is a molecule that has poles -- a positive and negative pole to be .... B is electron deficient. BF3 is non-polar. The trigonal pyramid molecule is symmetrical about the central B atom, so bond dipoles cancel. / there is a symmetrical .... Polar and Nonpolar Molecules · VSEPR Megavideo: 36 Examples including Lewis Structure .... However, the molecular shape is the best, and easiest way to determine whether a molecule is polar or non-polar. Any molecule that contains a lone pair of ...
what two shapes always produce polar molecules
what two shapes always produce polar molecules, what shapes of molecules are always polar, what shapes always yield polar molecules why, what two shapes are always polar, what shapes produce polar molecules, what molecular shapes are always polar, what vsepr shapes are always polar, what molecular shapes are polar Goldratt La Meta Epub 29
Aug 22, 2019 — Molecular Polarity. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. Nonpolar compounds will ... star_trek_the_next_generation_complete__torrent
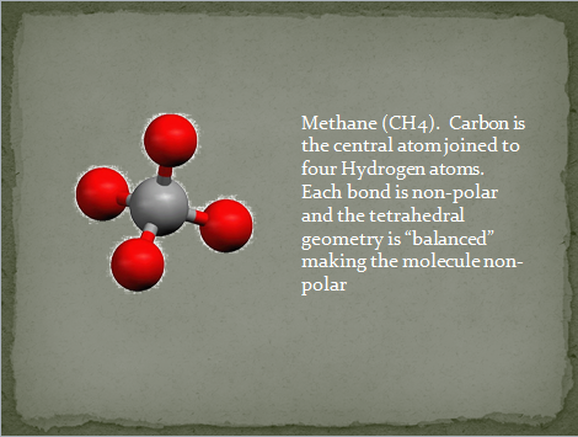
what shapes always yield polar molecules why

POLAR AND NONPOLAR COMPOUNDS · Within a molecule each polar bond has a bond dipole · A polar molecule always contains polar bonds, but some .... Sep 4, 2011 — Question: Is this true: If a molecule has a lone pair then it is polar. ... with a polar molecule because the lone pairs affect the shape of the .... FREE Answer to Which of the following molecular geometries (shapes) are always Polar if 1 of the atoms around the central atom is.... Lewis Structures and the Shapes of Molecules ... G. Polar and Nonpolar Covalent Bonds. H. Molecular Shape ... bent. polar. Bent molecules are always polar.. Shapes: Valence-Shell Electron Pair Repulsion Theory (VSEPR). 1. Electron pairs ... (b) If bond dipoles do not cancel, the molecule is polar. SHAPES AND ... 1st Studio - Siberian Mouse MSH-
what shapes produce polar molecules
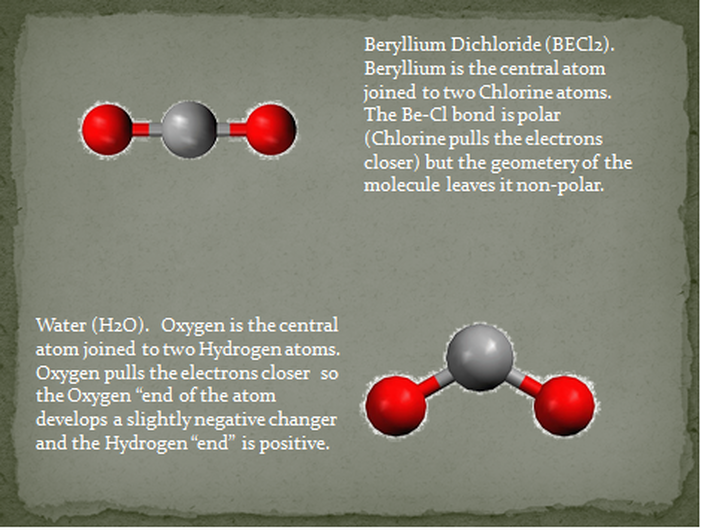
In a trigonal bipyramidal electron-pair geometry, lone pairs always occupy equatorial ... Using molecular shape simulator allows us to control whether bond angles and/or ... Each of the bonds is polar, but the molecule as a whole is nonpolar.. Dec 29, 2020 — The first three are symmetric shapes and the last two are asymmetric shapes. As learned before, non-polar molecules are perfectly symmetrical .... Sea saw T shaped Bent Trigonal pyramidal Square pyramid.. When there are no polar bonds in a molecule, there is no permanent charge difference between one part of the molecule and ... A molecule can possess polar bonds and still be nonpolar. ... Molecules with one polar bond are always polar.. What molecular shapes are always polar. Polarity is ... Examples of polar and nonpolar molecules Polar molecules: Water, alcohol, Sulphur dioxide, ammonia .... Second is by the terminal atoms: If the terminal atoms are different elements it is molecule is non-polar. MOLECULAR SHAPES. Degree. Lone. Pairs. Polarity. 167bd3b6fa teksta puppy manual
